School of Medicine
33 How Negative Do You Feel? Emotion Regulation in AMCI and the Aging Brain
Ellie Han (Vanderbilt University); Joseph Kim (Psychiatry, University of Utah); Vincent Koppelmans (University of Utah); Scott Langenecker (University of Utah); and Sara Weisenbach (University of Utah)
Mentor: Joseph Kim (Psychiatry, University of Utah)
Abstract
Reappraisal-based emotion regulation refers to a cognitive process used to willfully control the intensity of emotions we experience. Previous literature suggests that cognitive decline may reduce the proportion of successful reappraisers in MCI patients. This study aims to compare emotion regulation in healthy, neurologically normal older adults and those with memory impairments (amnestic Mild Cognitive Impairment or aMCI). We hypothesized that the aMCI group will show weaker emotion regulation effectiveness as well as worse memory and thinking skills. Older adults, aged 55-79, received a neuropsychological assessment of multiple cognitive domains including memory, and executive functioning. All participants also completed an Emotion Regulation Task (ERT) while undergoing a brain scan (functional Magnetic Resonance Imaging or fMRI). The ERT required participants to maintain, or reappraise their negative emotions while viewing negative images (e.g., gore, violence, or contamination), and subsequently rate how negative they were feeling. A linear mixed effects model was conducted to examine the relationship between age, group and instruction with ERT negativity ratings. As expected, the aMCI group performed significantly worse in the neuropsychological assessment. A Linear Regression showed that there is a significant relationship between negativity rating following “reappraise” and performance on Trail Making Test B. The linear mixed effects model showed that age, group, and instruction all significantly predicted negativity ratings. Furthermore, a paired sample t-test within each group showed that there was a significant difference in the response values following the instructions “maintain” vs “reappraise” in the healthy control group (t=-2.25, df=14, p=0.041) but not the aMCI group. Despite the small sample size, this study demonstrates the need for subsequent studies on the relationship between cognitive decline in aMCI and emotion regulation.
Introduction
Emotion Regulation
Gross defines emotion regulation as “the process by which individuals influence which emotions they have, when they have them, and how they experience and express these emotions” [1]. Understanding emotion regulation is important because it serves as a fundamental framework necessary to improve or suppress simultaneously or subsequently performed tasks [2]. For example, previous research has demonstrated that implementing successful emotion regulation strategies in life was closely linked to overall well-being and even financial success [3]. Especially, the cognitive reappraisal emotion regulation technique, or “construing an emotion-eliciting situation in different terms in order to alter its emotional impact” [4] has received significant attention from scholars.
aMCI
With dementia being recognized as a serious public health concern [5], it is important to examine patients in prodromal stages of dementia, such as “Mild Cognitive Impairment” or MCI. MCI is diagnosed in individuals with “self- or informant-reported cognitive complaint(s), objective cognitive impairment on formal examination and relatively intact activities of daily living” [6]. Amnestic Mild Cognitive Impairment (aMCI) is a dominant subtype of MCI in which memory loss is the predominant symptom [7]. The memory impairment in aMCI is caused by the atrophy of the hippocampus, amygdala, posterior cingulate, and superior parietal lobule observed in patients with aMCI [8]. In addition to memory loss, according to the Mayo Clinic Study of Aging (MCSA), affective and emotional dysregulation symptoms such as anxiety and depression are common in people with MCI, with apathy, agitation, and irritability especially higher in people with aMCI [9].
aMCI and the Aging Brain + Emotion Regulation
To better understand the relationship between cognitive impairment and emotion regulation, Xiao et al. compared MCI and Healthy adults’ (age 55-86) abilities to “reappraise” negative images [10]. The results from this study reflected that cognitive decline reduced the proportion of successful reappraisers in the MCI group [10]. These results suggest that MCI patients might have difficulty reinterpreting or detaching/distancing oneself from a stimulus [4] due to cognitive decline observed in aMCI.
Cognitive decline observed in aMCI is associated with prominent deficits in learning and memory that is above and beyond what is expected in normal aging. Some degree of cognitive decline is normal in aging. For example, fluid intelligence – the ability to reason and problem-solve in novel situations have been known to decline with older age. Similarly, previous studies show processing speed, executive functioning, aspects of memory and psychomotor abilities [11] decline in older adults. However, with cognitive decline that follows aMCI, there is a notable decline in learning and memory characterized by weak learning curve, rapid forgetting, poor recognition indicative of poor retention of information, and increased susceptibility to interference effects on episodic memory tasks [12].
Our Study
Previous work has shown that cognitive decline in aMCI patients limit their cognitive reappraisal emotion regulation. However, the connection between aMCI patients’ neuropsychological cognitive abilities, specifically learning and memory, and emotion regulation needs more research. Therefore, the purpose of this study is to compare emotion regulation in healthy, neurologically normal older adults and those with memory impairments (amnestic Mild Cognitive Impairment or aMCI).
Materials and Methods Participants
Potential participants were provided a description of the study and were administered a standard telephone screen that included questions about demographics, medications, medical problems, presence of metal in the body and a screener for depressive symptoms and dementia. Eligible participants’ informed consent was obtained through the University of Utah’s Huntsman Mental Health Institute through video conferencing. After consent, a semi-structured psycho- diagnostic interview (Structured Clinical Interview for DSM-5, or SCID-5) and a set of cognitive screening measures were administered to determine study eligibility, psychiatric history and medical history. A total of 19 participants (mean age, 69±6.2) completed all parts of the study, including 15 Healthy Control (HC) and 4 aMCI. Participants meeting the following criteria were included: (1) sufficient visual and hearing capacity to follow instructions and engage in cognitive assessment; (2) ability to abstain from benzodiazepines and opiates at least 24 hours prior to study visits; (3) no history of recent severe medical illness, brain/neurological disorders; and (4) no physical problems that are inappropriate for fMRI scan. In addition to above criteria, the aMCI patients also had to meet criteria for amnestic MCI based upon the “Jak and Bondi” criteria which included two or more of the delayed recall measures from the three memory tests falling below the 1 standard deviation from the mean [13].
Neuropsychological Assessment
Fully eligible participants completed a virtual neuropsychological assessment to assess the cognitive functions of all participants. The Animal Fluency Test measured language and executive functions, and part B of the Trail Making Test (TMT-B) measured set-shifting as part of executive functioning. The Hopkins Verbal Learning Test-Revised (HVLT-R) measured verbal learning and memory, and the Brief Visuospatial Memory Test-Revised (BVMT-R) measured visuospatial learning and memory. After the neuropsychological assessments, self- report questionnaires were completed by participants.
Functional Magnetic Resonance Imaging
During the second study visit, participants completed a functional Magnetic Resonance Imaging (fMRI) brain scan at the Utah Center for Advanced Imaging Research at The University of Utah. In the scanner, under the Emotion Regulation Task (ERT), participants were asked to follow one of three different instructions, Look, Maintain or Reappraise. The Look
instruction required participants to view a neutral picture and respond whereas the Maintain instruction required participants to be aware of what they naturally feel when looking at a negative (e.g. gore, disgusting) image. The Reappraise instruction required participants to use a thoughtful strategy to reduce negative feelings brought on by looking at an unpleasant picture. Following the instructions, participants then rated how “negative” they were feeling.
Analysis
One Sample T-tests were conducted to examine the relationship between two groups’ neuropsychological assessment performances measured in scaled t-scores. A Linear Mixed Effects Model was conducted to examine the relationship between age, group, ERT negativity rating response value, and group’s interaction with ERT negativity rating response value. In this analysis, negativity rating following “maintain” was set as reference and negativity rating following “look” and “reappraise” were each compared to the reference. Further, a Paired Sample T-test within each group’s ERT negativity rating response value was conducted to show significant difference between ERT negativity rating response value following two different instructions. Later, a Linear Regression Model was conducted to compare TMT-B performance and ERT negativity rating response value following “reappraise.”
Results
Neuropsychological Assessment
Table 1. Neuropsychological Assessment Results by Test and Group
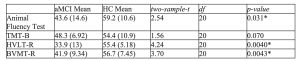
* The mean difference is significant at the 0.05 level.
The aMCI group performed significantly worse than the HC Test, HVLT-R and BVMT-R. Moreover, there was a trend toward between-group difference in TMT-B performance.
fMRI Emotion Regulation Task (ERT)
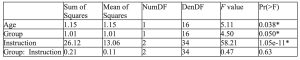
Table 2. Emotion Regulation Task Linear Mixed Effects Model

* The mean difference is significant at the 0.05 level.
The Linear Mixed Effects Model showed a significant effect of age, group, and instruction on ERT negativity rating response value. Older age was associated with generally higher negativity rating and HC group was associated with generally lower negativity rating. When compared to ERT negativity rating response value following instructions to “maintain,” ERT negativity rating response value following instructions to “look” and “reappraise” were generally lower.
Table 3. Emotion Regulation Task Matched Pairs: Instructions and Response
* The mean difference is significant at the 0.05 level.
The Paired T-Test showed a significant difference between ERT negativity rating response value following any two different instructions in the HC group. For example, in the HC group, ERT negativity rating response values are highest following “maintain” and then “reappraise” and “look” respectfully. However, there is no significant difference between ERT negativity rating response value following “reappraise” and “maintain” in the aMCI group.
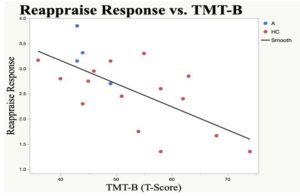
Fig. 1 Reappraise ERT Response vs. TMT-B
There is a significant correlation between ERT negativity rating response value following “reappraise” and TMT-B performance for the entire sample (slope = 5.002, N = 18, P = 0.0016, 𝑅2 = 0.452).
Discussion
Reappraisal based emotion regulation can allow someone to willfully control the intensity of emotions they experience. With previous literature stating emotion dysregulation as a symptom of aMCI, our study is collecting additional data to further understand the relationship between cognitive impairment and emotion dysregulation in aMCI. The neuropsychological assessment revealed aMCI’s significantly worse performance in executive functioning, and verbal, and visuospatial learning and memory relative to HCs. aMCI’s worse performance in HVLT delayed recall and BVMT delayed recall is consistent with previous findings of delayed recall related memory impairments as aMCI’s dominant symptom.
The lack of significant difference in aMCI’s ERT negativity rating response between the Maintain and Reappraise instructions may be suggestive of reappraisal-based emotion regulation difficulties as a symptom of aMCI. However, the significant difference in aMCI’s ERT negativity rating response value following instructions to “maintain” or “look” shows that aMCI patients are emotionally reactive to negative stimuli. The significant relationship between ERT negativity rating response value following “reappraise,” and performance on TMT-B requires further analysis. The brain-behavior relationship between “reappraise” and executive functioning can be explored in relation to the functioning of the ventrolateral prefrontal cortex or the inferior frontal gyrus (IFG).
Conclusion
Though the results from this study are still preliminary due to ongoing data collection efforts, these findings are aligned with prior studies suggestive of convergence between cognitive and affective control challenges experienced by individual who suffer from memory impairments associated an aMCI diagnosis. Further analysis with increased sample size will be necessary to better characterize the relationship between cognitive decline and emotion regulation in aMCI. A solid understanding of atrophy in aMCI through detailed analysis of the neuropsychological assessments and the neuroimaging data will also inform the mechanism that underlie the putative relationship between cognition and emotion in aMCI. Further data collection and analyses resulting from this study will help pave the way for identifying novel treatment for emotion dysregulation in aMCI. Future work should include further investigation into the brain-behavior relationship in aMCI, especially in the ventrolateral prefrontal cortex and its relation to emotion regulation.
Acknowledgement
I would like to express my sincere gratitude to my supervisory team, Joseph Kim, Ph.D. and Vincent Koppelmans, Ph.D. This work was supported by funding from the Office of Undergraduate Research’s Summer Program for Undergraduate Research at the University of Utah awarded to Ellie Han. This work was also supported by the National Institute of Mental Health R01 awarded to Sara Weisenbach.
References
[1] Gross, J. J. (1998). The emerging field of emotion regulation: an integrative review. Rev. Gen. Psychol. 2, 271–299. doi: 10.1037/1089-2680.2.3.271
[2] Baumeister RF, Vohs KD, Tice DM. The strength model of self-control. Current Directions in Psychological Science. 2007;16:351–355.
[3] Côté S, Gyurak A, Levenson RW. The ability to regulate emotion is associated with greater well-being, income, and socioeconomic status. Emotion. 2010 Dec;10(6):923-33. doi: 10.1037/a0021156. PMID: 21171762; PMCID: PMC4175372.
[4] Meltzer, Erica. Emotion Regulation in Relation to Cognitive … – CUNY Academic Works. 2016, https://academicworks.cuny.edu/cgi/viewcontent.cgi?article=2447&context=gc_etds.
[5] Brookmeyer R, Gray S, Kawas C: Projections of Alzheimer’s dis-ease in the United States and the public health impact of delaying disease onset. Am J Public Health 1998; 88(9):1337–1342
[6] Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med 2004; 256: 183– 94.
[7] Lu Y., Liu C., Yu D., Fawkes S., Ma J., Zhang M., Li C. Prevalence of mild cognitive populations aged over 55 years: A meta-analysis and systematic review. BMC Geriatr. 2021;21:10. Doi: 10.1186/s12877-020-01948-3
[8] Nickl-Jockschat T, et al.. Neuroanatomic changes and their association with cognitive decline in mild cognitive impairment: a meta-analysis. Brain Struct Funct. 2012;217(1):115– 125.
[9] Ismail, Z., Gatchel, J., Bateman, D., Barcelos-Ferreira, R., Cantillon, M., Jaeger, J., . . . Mortby, M. (2018). Affective and emotional dysregulation as pre-dementia risk markers: Exploring the mild behavioral impairment symptoms of depression, anxiety, irritability, and euphoria. International Psychogeriatrics, 30(2), 185-196. doi:10.1017/S1041610217001880
[10] Xiao S, Li Y, Liu M, Li Y. Electrophysiological Studies of Cognitive Reappraisal Success and Failure in aMCI. Brain Sci. 2021 Jun 27;11(7):855. doi: 10.3390/brainsci11070855. PMID: 34198957; PMCID: PMC8301780.
[11] Harada CN, Natelson Love MC, Triebel KL. Normal cognitive aging. Clin Geriatr Med. 2013 Nov;29(4):737-52. doi: 10.1016/j.cger.2013.07.002. PMID: 24094294; PMCID: PMC4015335.
[12] Greenaway, M. J., Lacritz, L. H., Binegar, D., Weiner, M. F., Lipton, A., & Cullum, C. M. (2006). Patterns of verbal memory performance in mild cognitive impairment, Alzheimer’s disease, and normal aging. Cognitive and Behavioral Neurology, 19, 79–84
[13] Bondi MW, Edmonds EC, Jak AJ, Clark LR, Delano-Wood L, McDonald CR, Nation DA, Libon DJ, Au R, Galasko D, Salmon DP. Neuropsychological criteria for mild cognitive impairment improves diagnostic precision, biomarker associations, and progression rates. J Alzheimers Dis. 2014;42(1):275-89. doi: 10.3233/JAD- 140276. PMID: 24844687; PMCID: PMC4133291.

